Translate this page into:
Brain stem auditory evoked potential response in neonates after recovery from hyperbilirubinemia: A neurophysiological approach to screening neuronal hearing loss
*Corresponding author: Ram Lochan Yadav, Department of Physiology, Chitwan Medical College, Bharatpur, Bagmati, Nepal. dr.ramlochan04@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Yadav RL, Yadav LK, Bhusal P, Timilsina S, Sapkota NK, Islam MN. Brain stem auditory evoked potential response in neonates after recovery from hyperbilirubinemia: A neurophysiological approach to screening neuronal hearing loss. Indian J Med Sci 2022;74:27-31.
Abstract
Objectives:
Neonatal hyperbilirubinemia leads to neurological damages including encephalopathy and hearing loss. This study aimed to screen and evaluate the hearing loss in neonates after recovery from hyperbilirubinemia using the Brainstem evoked response audiometry (BERA) test.
Materials and Methods:
This cross-sectional comparative study was conducted in Physiology Department at Chitwan Medical College, Nepal. It included 20 age and sex-matched neonates recently recovered from hyperbilirubinemia and 20 normal healthy controls. The external acoustic canals of subjects were checked for any blockage or collapse before BERA testing. The BERA recordings were performed after the neonate’s natural sleep following a standard lab protocol explained by Taylor’s Evoked Potential in Clinical Testing.
Results:
The BERA wave latencies were delayed with a higher number of case group neonates I (IL-75%, IR-80%), III (IIIL-70%, III R-80%), and V (VL-80%, VR-85%) than those of controls. The percentage of neonates with delayed interwave latencies was comparable between groups. The neonate’s hearing sensitivity assessed using the grades of hearing impairment by WHO revealed slight (threshold of hearing left ear [THL]-25% and threshold of hearing right ear [THR]-30%) and moderate (THL-40% and THR-35%) grades among cases whereas no impairment (THL-60% and THR-55%) in controls. The hearing thresholds were more in cases.
Conclusion:
The auditory pathway is highly sensitive to elevated serum bilirubin. BERA detects even a minute degree of hearing damage seen after complete treatment of hyperbilirubinemia. Therefore, BERA is a helpful tool in the early screening of hearing impairment in neonates. This improves prognosis by early management so that the neurosensory systems develop to their full extent and one can enjoy a normal social life.
Keywords
Brainstem auditory evoked potential test
Hearing evaluation
Neonatal hearing loss
Neonatal hyperbilirubinemia
INTRODUCTION
The normal speech and language development in healthy child depend on the well perception and processing of sound. The prevalence of hearing loss is 0.5–6/1000 neonates across the globe.[1] Nepal, a developing country, burdened with low-socioeconomic status and sociopolitical problem, has poorly developed medical system with less number of manpower, medicine, sophisticated instruments.[2] The hearing disability reported from 2011 (2068) census was 15.45% out of 1.94% total disability among Nepalese. The combined hearing loss and vision defect was reported in 1.48%, and among them 11.5% was reported with speech problem.[3] The prevalence of neonatal hearing impairment is supposed to be increased by 10–50 folds in infants at risk.[1]
Term-infants or newborns normally have relatively high level of bilirubin supposed to be due to: (1) lower activity and less number of glucuronosyltransferase enzymes needed for conversion of unconjugated bilirubin to conjugated bilirubin which is excreted into the gastrointestinal tract, (2) the shorter life span about 80–90 days of red blood cells in fetus compared to 100–120 days in adults, and (3) low rate of formation of urobilinogen from bilirubin by the intestinal flora, leading to greater absorption of bilirubin into the circulation pool.[4]
However, a chronic elevation of indirect serum bilirubin (>15 mg/dl) called Neonatal hyperbilirubinemia leads to detrimental health problem favoring morbidity and mortality.[5] The hyperbilirubinemia-induced dysfunction include wide neurological damage whose severity is determined by the duration and level of bilirubin exposure to the central nervous system. Kernicterus, acute bilirubin encephalopathy, and specific neural pathway dysfunction including the auditory pathway are some common neurological hazards caused by hyperbilirubinemia. The bilirubin at high level has the highest sensitivity in damaging the auditory system affecting the hearing pathway and predominantly the retro-cochlear structures such as the brainstem auditory nuclei, inferior colliculi, spiral ganglion neurons, and auditory nerve fibers.[6-8] The auditory damage caused by hyperbilirubinemia has long-term and permanent effects if quick management is not done. Moreover, language development is severely impaired since it largely dependent on healthy auditory function.[9]
Therefore, it is quite pertinent to identify and evaluate the hearing loss by the hyperbilirubinemia to prevent such neurological complications. Some routine investigations are used for screening and early detection of defect in developed countries. It is quite impossible to test the neonates and infants reliably by conventional audiometric methods using behavior observation, free field audiometry, visual reinforcement audiometry, play audiometry, etc. Thus, for the reliable results, the test not requiring the active co-operation of patient is needed. Brainstem evoked response audiometry (BERA) is such a test. BERA is an easy, non –invasive, objective test to explore the integrity along the central course of auditory pathways through the 8th cranial nerve, pons, and midbrain for early identification of hearing impairment in children and neonates.[10] It can be used as a screening test in newborns, infants, and other uncooperative subjects. In this study, threshold estimation was done using BERA in high-risk neonates to detect hearing loss and also to find the degree of impairment. It is obvious that early detection of hearing impairment early in life is required to improve the prognosis so that one can be benefitted fully of the developing sensory systems and enjoy normal social circumstances. Therefore, this study aimed at assessing the abnormal BERA waves and threshold of hearing in neonates recently recovered from hyperbilirubinemia and compare with healthy neonates who had never hyperbilirubinemia.
MATERIAL AND METHODS
This cross-sectional comparative study, conducted in the Department of Clinical Physiology, Chitwan Medical College (CMC), Nepal, included twenty male neonates diagnosed with hyperbilirubinemia[11] (total serum bilirubim [TSB] >15 g/dl) and have gone through the appropriate treatment as the study group and 20 age and sex-matched non-icteric babies who never had hyperbilirubinemia in the past as control group. All the subjects were recruited from the Department of Pediatrics, CMC. The study group neonates were subjected to BERA tests after one week of discharge after treatment of neonatal jaundice by phototherapy and medication. At the time of BERA test, all the neonates (both the study and control groups) had normal level of total serum bilirubin (TSB ≤8 mg/dl included in this study). We could not perform BERA tests soon after diagnosis made as neonatal jaundice and in follow-up because of lack of trend as screening test for hearing using BERA by Clinicians in Nepal. This also could be due to poor financial circumstances of the parents. Therefore, we conducted BERA test both in the study and control groups neonates but only at once.
Preterm neonates and neonates born with a complicated pregnancy, abnormal conditions like- birth asphyxia, meningitis, septicemia, congenital abnormalities such as cleft lips and palate or babies on mechanical ventilator, low birth weight, ototoxic medication, and abnormal otoscopy were excluded from this study. The control group was comprised of neonates who had never hyperbilirubinemia in the past.
Neonate’s mothers were well explained about the test procedure and further Informed written consent was obtained from all of them. The neonates were screened for the study based on above mentioned inclusion and exclusion criteria set for the study. Room temperature of the laboratory was maintained at the thermoneutral zone, that is, 28°C ± 2°C. The required setup was checked before starting the test. Prior to commencing the BERA test, the external acoustic canal was examined to exclude any blockage of the meatus by wax or collapsing the acoustic lumen. Both ears were tested and the BERA recordings began after the neonates had natural sleep. The re-cording machine for BERA was Neuropack MEB-2300, Nihon Kohden, Japan. Since the electrodes are placed on the scalp, the patient’s mother was instructed to have shampoo/soap bath of the scalp of baby and keep oil and grease-free before coming for investigation. BERA recording was done with standard electrode placement which included a non-inverting electrode (positive) over the vertex of the head, and inverting electrodes (negative) over the mastoid prominence, as explained by Taylor.[12] The earthing (ground) electrode, important for proper functioning of preamplifier, is placed over the middle forehead. This earthing electrode is important for proper functioning of preamplifier. The inter-electrode impedance was checked and kept below 5 kΩ during the test. The sweep velocity adjusted was 10 ms. The earphone-based click acoustic stimuli given at the rate of 10/second alternating in polarity to each ear alternately at an intensity of 80 dB. The noses, if any, were removed and electrical activities were filtered and averaged to 2000 responses. The variables recorded were: Mean latency time of waves I, III, and V, Interwave latencies of waves I-III, I-V, and III-V. The absence of waves, if any, was noted to identify the neurological defects along the hearing pathway. Further, to identify the threshold of hearing, 2000 recordings were averaged decreasing in steps of 5 dB starting from 99 dB. The graph structure was noted until wave V is no longer identifiable. The minimum intensity when wave V is detected is taken as the hearing threshold for that neonate. In our study, 30 dB is taken as cutoff for normal hearing threshold. All the subjects were tested by the first author of this study to avoid the technical errors might be encountered when testing done by different technicians. The hearing loss was quantified as mild, moderate, severe, and profound. The neonate’s hearing sensitivity was assessed based on the following grades of hearing impairment by the WHO [Table 1].[13]
| Grade of hearing impairment | Threshold level (dB) |
|---|---|
| No impairment (0) | 25 or better |
| Slight impairment (1) | 26–40 |
| Moderate impairment (2) | 41–60 |
| Severe impairment (3) | 61–80 |
| Profound impairment (4) | >80 |
This study was conducted according to the guidelines of the Declaration of Helsinki and approval was taken by the Institutional Review Ethical Committee, CMC, Nepal prior to study.
Statistical analysis
The SPSS (version 20; IBM Corporation, Armonk, NY, USA) was used for data analysis. The descriptive tests were applied in both the case and control groups for BERA variables. The threshold of hearing was compared between the groups using descriptive test. Descriptive analysis was done because this was a cross-sectional comparative and non-probability type of study.
RESULTS
All the neonates included in the study were male with the mean age of babies was 23.18 ± 1.12 days, mean weight 3.6 ± 0.57 kg, mean gestational age 38.52 ± 1.46 weeks and mean serum Bilirubin 6.81 ± 0.17 mg/dl. The latency of each wave was found to be prolonged even after treatment of hyperbilirubinemia. The BERA wave latencies delayed in high number of neonates I (IL-75%, IR-80%), III (IIIL-70%, III R-80%), and V (VL-80%, VR-85%) among cases than those of the controls [Table 2]. The percentages of neonates with prolonged inter-peak latencies were comparable between the groups. The neonate’s hearing sensitivity assessed based on the grades of hearing impairment by the WHO revealed slight (threshold of hearing left ear [THL]-25% and threshold of hearing right ear [THR]-30%) and moderate (THL-40% and THR-35%) grades of hearing impairment among cases which indicated that every neonate even after treatment of hyperbilirubinemia had some degree of hearing defect. In controls, high percentage revealed no any impairment (THL-60% and THR-55%) and few with slight impairment (THL40% and THR-45%) [Table 3 and Figure 1]. On comparison, the threshold of hearing was higher in neonates after treatment of hyperbilirubinemia (cases). None of the cases were identified without hearing impairment and 35–40% of cases have moderate impairment of hearing defined by the WHO.
| BERA waves | BERA waves in left (L) and right (R) ears | Delayed BERA in neonates case group | |
|---|---|---|---|
| Case group (n=20) (%) | Control group (n=20) (%) | ||
| I wave latency | IL | 75 | 10 |
| IR | 80 | 20 | |
| III wave latency | IIIL | 70 | 10 |
| IIIR | 80 | 10 | |
| V wave latency | VL | 80 | 5 |
| VR | 85 | 15 | |
| I-III inter-peak latency | I-IIIL | 30 | 30 |
| I-IIIR | 35 | 35 | |
| I-V inter-peak latency | I-VL | 20 | 15 |
| I-VR | 30 | 20 | |
BERA: Brainstem evoked response audiometry
| Grade of impairment | Variables | Cases (n=20) (%) | Controls (n=20) (%) |
|---|---|---|---|
| No impairment (0) | THL* | 0 | 60 |
| THR* | 0 | 60 | |
| Slight (1) | THL | 25 | 40 |
| THR | 30 | 45 | |
| Moderate (2) | THL | 40 | 0 |
| THR | 35 | 0 | |
| Severe impairment (3) | THL | 0 | 0 |
| THR | 0 | 0 | |
| Profound impairment (4) | THL | 0 | 0 |
| THR | 0 | 0 |
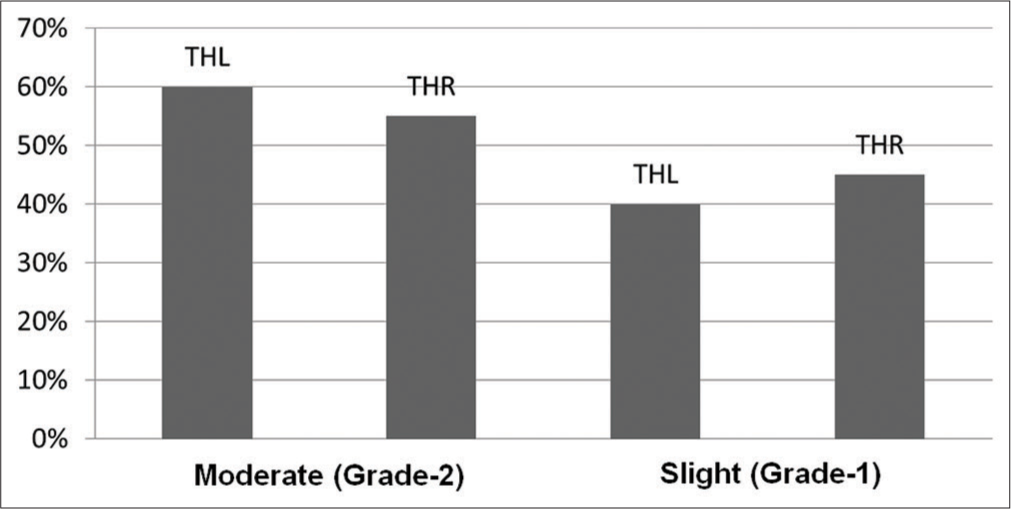
- Grade of hearing impairment (WHO) in cases (n=20).
DISCUSSION
The elevated serum unconjugated bilirubin in neonates easily passes through the blood brain barrier and could develop acute encephalopathy. The chronic hyperbilirubinemia causes abnormal precipitation of unconjugated and free bilirubin in various significant brain areas causes encephalopathy with several detrimental health effects including neuronal functions. Neonatal hyperbilirubinemia is known to be a significant risk factor for auditory dysfunctions.[5,14] With recent advances in medical field, the hyperbilirubinemia- induced hearing damages in the neonate are studied using brainstem auditory evoked response BERA, the simple, reliable, objective method to study along the course of auditory pathway.
In this study, the BERA was abnormal in 70–80% of neonates who had just recovered from hyperbilirubinemia, which is comparable to outcomes in other studies.[15] The latencies of BERA waves I, II, and V were prolonged. These findings are in consonance to findings of other authors reporting delayed BERA at the time of discharge.[16,17] Vinodh et al. reported that BERA abnormalities (including interwave I-III and I-V abnormality) were noted in 18 (17.6%) ears even at the time of discharge.[18] These findings also support our findings with the presence of abnormal BERA response in neonates after treatment of hyperbilirubinemia but the percentage of neonates with abnormal BERA was less than those in our study. Several studies[19,20] reported prolongation of BERA wave II, III, and V latencies similar to findings in this study but in contrast to our study they reported with normal BERA I wave latency.
After treatment of neonatal jaundice, BERA abnormalities in form of prolonged interwave interval were comparable between the groups. These findings are in line with the previous report indicating prolonged interwave interval in seven cases while the latencies of various waves had normalized in most cases.[21] In our study, the BERA waves I, III, and V were prolonged in cases than the control group. This might be either due to long-term effect of hyperbilirubinemia or the damaged auditory pathway was still in recovery process. The BERA abnormalities persistence in some cases after treatment could be due to irreversible damage caused by axonal degeneration and/or loss of myelin rather than hair cell loss.[22] Deorari et al.[23] reported similar findings while other authors[24] reported that on follow-up all neonates with abnormal BERA showed a recovery back to normal. Such transient bilirubin toxicity or the transient brainstem encephalopathy postulated the hypothesis of improved brain functions due to removal of bilirubin from the brainstem because of phototherapy and/or exchange transfusion.
In this study, the cases had raised threshold of hearing. The neonate’s hearing sensitivity assessed based on the grades of hearing impairment by the WHO revealed slight (THL-25% and THR-30%) and moderate (THL-40% and THR-35%) grades of hearing impairment among cases which indicated that every neonate even after treatment of hyperbilirubinemia had some degree of hearing defect indicated by raised threshold. In controls, a high percentage revealed no any impairment (THL-60% and THR-55%) and few with slight impairment. Similar to our findings, Soni et al[23] reported that 8 (26.6 %) cases had raised threshold even at discharge. Sharma et al.[20] report reported that abnormal outcomes were obtained in 73.3% of cases and the irreversible abnormality in the follow-up tracings in 23.3% of the study group. These findings are comparable to our findings.
CONCLUSION
The study showed a high percentage of hearing impairment through abnormal BERA responses in neonates discharged after treatment of hyperbilirubinemia. They had also raised the threshold of hearing. The study brings some important issues for further evaluation in follow-up of hyperbilirubinemia cases. The clinicians should have aware of the ideal age for screening these infants, as early screening results in abnormal BERA in a larger number of neonates. Further, the neonates should be screened for hearing using BERA tests soon after diagnosis of hyperbilirubinemia and in follow-up after treatment of hyperbilirubinemia to find the reversibility auditory damage, though this could be limitations of this study. Auditory pathway is highly sensitive to high serum bilirubin. BERA detects even a minute degree of hearing damage seen after treatment of hyperbilirubinemia. Therefore, BERA could be a helpful tool in the early screening of hearing impairment in neonates. This improves prognosis and early management so that the neurosensory systems develop to its full normal extent and the child can be benefited with normal social development.
Limitations of the study
It would be better if we could include a large number of samples for the study. The BERA evaluation could be done at the time of diagnosis of hyperbilirubinemia and in follow-ups to evaluate immediate change in auditory functions. These limitations were due to poor financial circumstances raised due to lack of any funds for this study.
Acknowledgment
We thank the parents of neonates being available for the tests in time. We acknowledge the Pediatricians for their support by appropriate counseling to neonate’s parents.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- The economics of screening infants at risk of hearing impairment: An international analysis. Int J Pediatr Otorhinolaryngol. 2012;76:212-8.
- [CrossRef] [PubMed] [Google Scholar]
- Nepal Population Report 2011 Published by: Government of Nepal Ministry of Health and Population. 2011. Population Division Ramshahpath, Kathmandu, Nepal. Available from: https://www.mohp.gov.np [Last accessed on 2021 Feb 27]
- [Google Scholar]
- National Population and Housing Census 2011 (National Report) Government of Nepal. Vol 1. Nepal: National Planning Commission Secretariat, Central Bureau of Statistics Kathmandu; 2011.
- [Google Scholar]
- Movement of bilirubin and bilirubin conjugates across the placenta. Pediatrics. 2007;119:1032-3.
- [CrossRef] [PubMed] [Google Scholar]
- Bilirubin toxicity in the developing nervous system. Pediatr Neurol. 2003;29:410-21.
- [CrossRef] [PubMed] [Google Scholar]
- Association between peak serum bilirubin and neurodevelopmental outcomes in extremely low birth weight infants. Pediatrics. 2003;112:773-9.
- [CrossRef] [PubMed] [Google Scholar]
- To find the prevalence of hearing impairment by brainstem evoked response audiometry in high risk neonates attending a tertiary care hospital in their follow up visit. Sch J App Med Sci. 2018;6:4498-502.
- [Google Scholar]
- Early intervention and language development in children who are deaf and hard of hearing. Pediatrics. 2000;106:E43.
- [CrossRef] [PubMed] [Google Scholar]
- New Handbook of Auditory Evoked Responses (1st ed). London, United Kingdom: Pearson; 2007.
- [Google Scholar]
- Jaundice and hyperbilirubinemia in the newborn In: Nelson Text Book of Pediatrics Vol Part XI. (16th ed). 2001. p. :513-9.
- [Google Scholar]
- Evoked potential in pediatrics In: Edinburg AM, ed. Evoked Potential in Clinical Testing (2nd ed). London: Churchill Livingstone; 1993. p. :489-521.
- [Google Scholar]
- WHO: Prevention of Blindness and Deafness. 2013. Geneva: World Health Organization; Available from: http://www.who.int/pbd/deafnee/hearing_impairment_grades/en [Last accessed on 2021 Feb 27]
- [Google Scholar]
- Brainstem auditory evoked response in neonatal neurology. Semin Fetal Neonatal Med. 2006;11:444-51.
- [CrossRef] [PubMed] [Google Scholar]
- Auditory brainstem responses in patients with neonatal hyperbilirubinemia and bilirubin encephalopathy. Brain Dev. 1989;11:297-301.
- [CrossRef] [Google Scholar]
- Identification of neonatal hearing impairment: Characteristics of infants in the neonatal intensive care unit and well-baby nursery. Ear Hear. 2000;21:373-82.
- [CrossRef] [PubMed] [Google Scholar]
- Is auditory brainstem response a bilirubin neurotoxicity marker? Am J Otolaryngol. 1998;19:232-6.
- [CrossRef] [Google Scholar]
- Reversibility of brainstem evoked response audiometry abnormalities at 3 months in term newborns with hyperbilirubinemia. Indian Pediatr. 2014;51:134-5.
- [CrossRef] [PubMed] [Google Scholar]
- Brainstem auditory evoked response in newborn with hyperbilirubinemia. Indian J Pediatr. 1998;35:513-8.
- [Google Scholar]
- Brainstem evoked response audiometry (BAER) in neonates with hyperbilirubinemia. Indian J Pediatr. 2006;73:413-6.
- [CrossRef] [PubMed] [Google Scholar]
- Brainstem evoked response audiometry (BERA) in neonates with hyperbilirubinemia. Indian J Otolaryngol Head Neck Surg. 2016;68:334-8.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical and pathological correlates of brainstem auditory response abnormalities. Neurology. 1977;27:316-25.
- [CrossRef] [PubMed] [Google Scholar]
- One year outcome of babies with severe neonatal hyperbilirubinemia and reversible abnormality in brainstem auditory evoked responses. Indian Pediatr. 1994;31:915-21.
- [Google Scholar]
- Auditory brainstem response and clinical follow-up of high-risk infants. Pediatrics. 1989;83:385-92.
- [Google Scholar]






