Translate this page into:
Cytological diagnosis of soft-tissue lesions: A study of variation in site of occurrence
*Corresponding author: Sompal Singh, Department of Pathology, Hindu Rao Hospital, New Delhi, India. sompal151074@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Sharma SK, Prakash P, Kalra R, Naeem R, Jahan A, Sarin N, et al. Cytological diagnosis of soft-tissue lesions: A study of variation in site of occurrence. Indian J Med Sci 2021;73:331-4.
Abstract
Objectives:
Fine-needle aspiration cytology is a useful diagnostic tool for soft-tissue lesions, which helps in further management of patient. Although, various soft-tissue lesions have preference for particular site of human body, yet usefulness/role of site in cytological diagnosis in the published literature is not very well highlighted. In this study, our aim was to analyze the location of soft-tissue lesions as encountered in cytology practice.
Material and Methods:
Details of soft-tissue lesions who underwent fine-needle aspiration in a period of 12 months were analyzed. Age, gender, and site of soft-tissue lesion were noted. All the soft-tissue cases were divided into two categories. Group 1 – soft-tissue lesions situated at elbow, forearm, wrist, and hand for upper extremities; and knee, lower leg, ankle, and foot for lower extremities. Group 2 – soft-tissue lesions occurring at other sites. Pearson’s Chi-square test and significance of difference between proportions were analyzed to assess the significance.
Results:
A total of 928 cases were reported over a period of 12 months. Soft-tissue lesions in Group 1 and Group 2 were 172 and 756 cases, respectively. All the soft-tissue lesions except ganglion cyst (GC) and giant cell tumor (GCT) of tendon sheath are more prevalent in Group 2. There was a statistically significant association (P < 0.001) between cytological diagnosis of soft-tissue lesion and site of lesion. The difference in occurrence of epidermal inclusion cyst, GC, and GCT of tendon sheath between Groups 1 and 2 was found to be statistically significant (P < 0.001).
Conclusion:
We found significant difference in occurrence of some of the soft-tissue lesion at distal extremity as compared to other location. This may be helpful in arriving at a proper cytological diagnosis.
Keywords
Soft tissue
Fine-needle aspiration
Location
INTRODUCTION
Fine-needle aspiration cytology (FNAC) is a universally accepted, simple, minimally invasive, economical, rapid, and useful diagnostic tool.[1] Soft-tissue lesions include non-neoplastic and neoplastic lesion (tumor).
Soft-tissue lesions are challenging for the cytopathologist due to their biological heterogeneity; and broad and overlapping morphological features.[2] Nowadays, FNAC is used as primary diagnostic tool in the diagnosis of soft-tissue lesions.[3]
Soft-tissue tumors are the mesenchymal proliferations that are found in the extraskeletal, non-epithelial tissues of the body, except the viscera, outer layer of brain, and lymphoreticular system.[4] Soft-tissue tumor can be benign or malignant.
In general, benign soft-tissue tumors outnumber their malignant counterparts by ratio of about 100:1 in hospital population with annual incidence of 300/100,000 population.[4,5] The diagnostic accuracy of FNAC for soft-tissue tumors differentiating between benign and malignant lesions is very high.[6] However, the lack of tissue architecture in FNAC smear makes it more difficult to categorize correctly the soft-tissue lesions.[7]
FNAC is being increasingly used for cutaneous and subcutaneous lesions as it decreases the incidence of unnecessary surgical excisions and/or biopsy.[8] It has been suggested that cytological categorization of soft-tissue tumor as benign, intermediate, and malignant lesions would help in management of the patient.[9] The prognosis of malignant soft-tissue tumor is unpredictable and they may be very aggressive if not diagnosed early.
Soft-tissue lesion can occur at different sites of the body, although various soft-tissue lesions have preference for particular site of human body. Although, the site of lesion during FNAC is very helpful in cytological diagnosis of soft-tissue lesions, yet the focus in this area in the published literature is subtle. There are number of studies in literature[8,10] describing the usefulness of FNAC in soft-tissue lesions, however, the site of soft-tissue lesions has not been much highlighted in cytology literature. The aim of our present study was to analyze the location of soft-tissue lesion in cytological diagnosis.
MATERIAL AND METHODS
The present study was a retrospective study conducted in cytopathology section of a tertiary care medical college and hospital. Record of soft-tissue lesions was retrieved for the period of 12 months (January 2019–December 2019). Following details of patient were noted including age, gender, and site. All the soft-tissue cases were divided into two categories. Group 1 – soft-tissue lesions situated at elbow, forearm, wrist, and hand for upper extremities; and knee, lower leg, ankle, and foot for lower extremities. Group 2 – soft-tissue lesions occurring at other sites.
Statistical analysis
To assess the statistical significance of association between cytological diagnosis and site of lesion, Pearson’s Chi-square test was used. To assess the significance of difference in proportion of cases in Groups 1 and 2, tests for significance of difference between proportions were used. P < 0.05 was considered as statistically significant. Open sourced statistical software “R” (vesion-4.0) in combination with R studio (version-1.1.463) was used for statistical analysis.
RESULTS
There were 928 cases of soft-tissue FNAC done over a period of 12 months. The age of patient ranged from 1 month to 85 years with a mean age of 31.6 years (SD = 14.8 years). Total number of male and female was 524 and 404, respectively, with a female-to-male ratio of 1:1.3. Soft-tissue lesions in Group 1 were 172 cases and in Group 2 were 756 cases, respectively.
Distribution of soft-tissue lesion according to location in terms of Group 1 and Group 2 is shown in Table 1. None of our case had histological follow-up, including cell block preparation. Among Group 2 lesions, the most common site of occurrence was back (132 cases, 17.46%), followed by abdomen (95 cases, 12.56%). The other common sites among Group 2 were scalp (54 cases, 7.14%) and chest (52 cases, 6.87%). In the present study, all the soft-tissue lesions except ganglion cyst (GC, Figure 1b) and giant cell tumor (GCT, Figure 1c) of tendon sheath are more prevalent in Group 2. All the cases of GCT of tendon sheath were located at distal extremities. About 94.7% of cases of GC occurred at distal extremities. Among the other common lesions, 17.5% of cases of lipoma, 9.4% of cases of epidermal inclusion cyst, and 35.3% of cases of spindle cell mesenchymal neoplasm (SCMN) (possibly benign) occurred at distal extremities. Tuberculosis (TB) was seen in both Group 1 and Group 2, however, 6.9% of cases occurred at distal extremities. There was a statistically significant association (P < 0.001) between cytological diagnosis of soft-tissue lesion and site of occurrence. The significance of difference between proportion in Groups 1 and 2 of individual entity was assessed. The difference in occurrence of epidermal inclusion cyst, GC, and GCT between Groups 1 and 2 was found to be statistically significant (P < 0.001).
| Category | Group 1 (%) |
Group 2 (%) |
Total (%) |
Significance of difference |
|---|---|---|---|---|
| Spindle celled mesenchymal neoplasm (possibly benign) | 12 (6.97) |
22 (2.9) |
34 (3.66) |
0.010 |
| Spindle celled mesenchymal neoplasm (possibly borderline) | 0 (0) |
1 (0.13) |
1 (0.1) |
0.631 |
| Calcinosis cutis | 0 (0) |
1 (0.13) |
1 (0.1) |
0.631 |
| Cystic lesion | 6 (3.48) |
24 (3.17) |
30 (3.2) |
0.833 |
| Epidermal inclusion cyst | 21 (12.2) |
203 (26.85) |
224 (24.14) |
<0.001 |
| Ganglion cyst | 18 (10.46) |
1 (0.13) |
19 (2.0) |
<0.001 |
| Giant cell tumor of tendon sheath | 4 (2.3) |
0 (0) |
4 (0.43) |
<0.001 |
| Granulomatous lesion | 0 (0) |
12 (1.58) |
12 (1.3) |
0.096 |
| Inflammatory lesion | 15 (8.7) |
52 (6.87) |
67 (7.22) |
0.400 |
| Lipoma | 62 (36.0) |
292 (38.6) |
354 (38.15) |
0.528 |
| Malignant neoplasm | 1 (0.58) |
5 (0.66) |
6 (0.65) |
0.904 |
| Inadequate material for opinion | 29 (16.86) |
89 (11.77) |
118 (12.7) |
0.070 |
| Parasitic lesion | 0 (0) |
6 (0.79) |
6 (0.65) |
0.242 |
| Reparative change | 1 (0.58) |
4 (0.53) |
5 (0.54) |
0.936 |
| Skin adnexal neoplasm | 0 (0) |
13 (1.72) |
13 (1.4) |
0.083 |
| Tubercular lesion | 2 (1.16) |
27 (3.57) |
29 (3.1) |
0.101 |
| Suggestive of vascular neoplasm/lesion | 1 (0.58) |
4 (0.53) |
5 (0.54) |
0.936 |
| Total | 172 | 756 | 928 | - |
Bold indicates significant p value
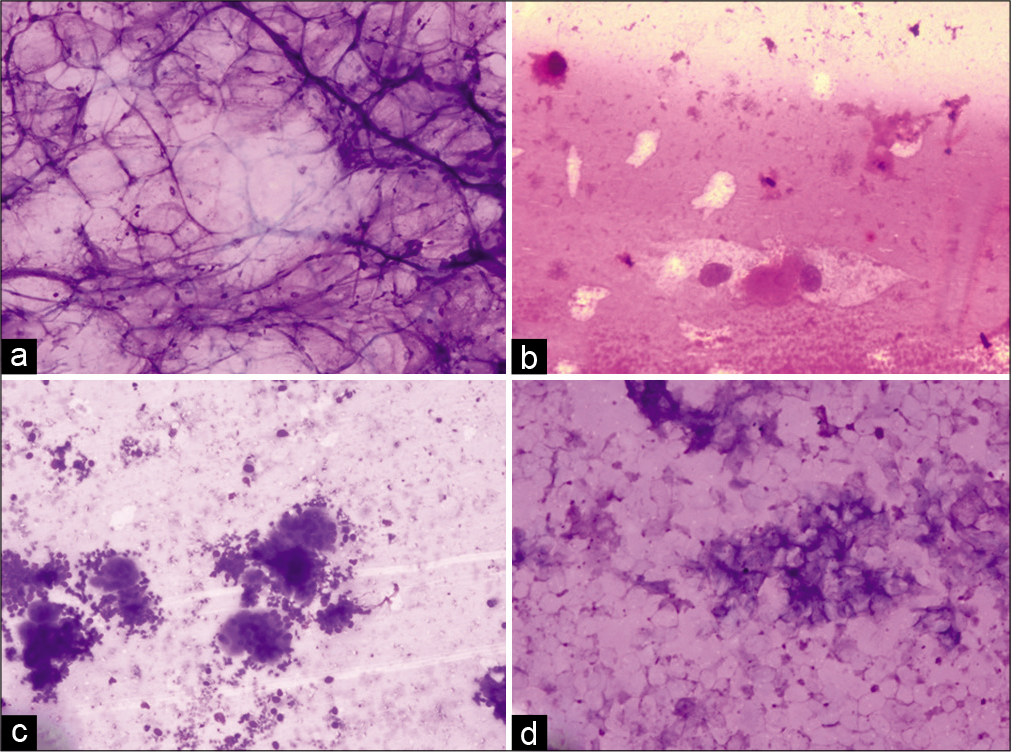
- Photomicrograph showing lipoma composed of adipocytes (a, ×40, MGG), ganglion cyst showing scattered cells with abundant cytoplasm (b, ×40, MGG), giant cell tumor of tendon sheath showing multinucleated giant cells and stromal cells (c, ×40, MGG), and epidermal inclusion cyst showing scattered anucleate squames (d, ×40, MGG).
There were 29 cases in Group 1 and 89 cases in Group 2 which had insufficient cytological material for reporting. In all of these cases, repeated attempts of FNAC were performed without any improvement in cellularity.
In Group 1, there was one malignant case which was suggestive of adenocarcinoma. In Group 2, there were five cases of malignancy, of which three were suggestive of adenocarcinoma, one each was suggestive of sarcoma and malignant adnexal neoplasm.
We encountered six cases of parasitic lesions, all of which were in Group 2. Of the parasitic lesion, four were cytologically diagnosed as cysticercosis, while two showed presence of microfilaria.
DISCUSSION
The diagnosis of soft-tissue lesions can be obtained by FNAC, core biopsy, or open biopsy with each having their own advantages and disadvantages. As compared to core/open biopsy, fine-needle aspiration procedure is well tolerated by patients as risk of complication is negligible. Moreover, multiple sampling can be done by multiple passes of fine needle from different parts of lesion, on the contrary, a single core biopsy does limited sampling.[11,12]
In our study, 928 cases of soft-tissue lesions underwent fine-needle aspiration over a period of 12 months. The age of patient ranged from 1 month to 85 years with a mean age of 31.6 years. Our study showed that maximum number of cases occur in 20–30 years age group followed by 30–40 years age group. There was male predominance with female-to-male ratio of 1:1.3. Similarly, in a study done by Dash et al.[8] on 712 cases of soft-tissue lesions, there was slight male preponderance with a female-to-male ratio of 1:1.4.
Lipomas are benign soft-tissue tumor of adipocyte origin. They are usually subcutaneous and may be solitary or multiple. In our study, lipoma was most common cytological diagnosis (354 cases out of 928, 38.14%). Dey et al.[13] observed similar result with about 40% of lipomas among all soft-tissue tumor and tumor like conditions they studied. Similarly, Siddiqie Hassan Adil et al.[10] found about 60% of soft-tissue lesions as lipomas. According to literature, histologically most common location of lipoma is trunk and shoulder followed by proximal part of extremities.[14] Kransdorf et al.[15] studied 1453 lipomas out of which only 151 cases, 10.39%, were located at distal extremities. In terms of absolute numbers, we found that similar results with number of lipoma cases in Group 2 were much more than in Group 1. However, among Group 2, lipomas were 38.62% as compared to 36.04% in Group 1. This difference was statistically insignificant (P = 0.528). Hence, among all soft-tissue lesions occurring at distal extremity, chances of lesion being lipoma is same as those occurring at proximal extremity or trunk area.
Epidermal inclusion cyst is another common cutaneous lesion. Second most common diagnosis in our cases of soft-tissue lesions was epidermal inclusion cyst (224 cases out of 928 cases, 24%). They may occur anywhere in the body but the face, scalp, neck, and trunk are the most common site.[16,17] Very occasionally, they occur on palm or soles.[17] In our study, epidermal inclusion cyst was seen in both the groups. Among cases seen at distal extremity, they comprised about 12.2% as compared to 26.85% among cases occurring at proximal extremity and trunk. This difference was statistically significant (P < 0.001).
We encountered 67 cases (7.22%) diagnosed as non-specific inflammatory lesion on cytology. The proportion of inflammatory lesion in Group 1 (8.7%) and in Group 2 (6.87%) was similar (P = 0.400).
In our study, there were 35 cases of SCMN, out of which 34 were benign appearing on cytology. Benign SCMN occurred in both Group 1 and Group 2. In Group 1, it comprised 6.97% of cases as compared to 2.9% of cases in Group 2.
TB is the most common chronic infectious disease in developing countries. TB can occur at any organ/anatomical site of the body having varied presentation. Among extrapulmonary sites, cervical lymph node is the most common sites for TB.[18,19] In the present study, among Group-2, 27 cases (3.57%) were of TB as compared to 2 cases (1.16%) among Group 1. Hence, although cases were seen in both the groups, chances of soft tissue being of tubercular etiology, are more in proximal extremity and trunk as compared to distal extremity.
GC is a degenerative lesion involving musculoskeletal system.[20] They are usually encountered in distal extremities.[21] In our study of all the GC, most cases occurred in distal extremities. Only one case was in Group 2, which was 0.13% of all cases in Group 2. The difference in proportion of cases in Group 1 and Group 2 was statistically significant (P < 0.001).
In our study, GCTs of tendon sheath were seen only at distal extremity. This is consistent with studies in English literature. Kransdorf et al.[15] studied 731 cases of GCT out of which 587 cases (80.3%) were located at distal extremities.
In our study, skin adnexal lesions (13 cases) and parasitic lesions (6 cases) were found in Group 2 only, though there are reports of parasitic lesions and skin adnexal neoplasms occurring in distal extremities.[22,23]
CONCLUSION
In the present study, we found that there was a significant difference in occurrence of some of the soft-tissue lesions at distal extremity as compared to other location. Although not diagnostic, information of site of lesion may aid in approach toward a proper cytological diagnosis of the soft-tissue lesions and hence their subsequent management.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Study of fine aspiration cytology in lymphadenopathy with special reference to Acid Fast Staining in cases of Tuberculosis. JK Sci. 2005;1:1-4.
- [Google Scholar]
- Sarcomas of soft tissues In: Vita VD, Hellman S, Rosenberg SA, eds. Cancer: Principles and Practice of Oncologyl (3rd ed). Philaldelphia, PA: JB Lippincott; 1989. p. :1345-98.
- [Google Scholar]
- Cytopathology of Bone and Soft Tissue Tumors Oxford; Oxford University Press; 2002.
- [Google Scholar]
- Fine needle aspiration cytology of primary soft tissue tumors. Morphologic analysis of the most frequent types. Acta Cytol. 1992;36:905-17.
- [Google Scholar]
- Fine needle aspiration cytology vs. needle core biopsy of soft tissue tumors. A comparison. Acta Cytol. 1994;38:381-4.
- [Google Scholar]
- Fine needle aspiration cytology of soft tissue tumors. Acta Cytol. 2000;44:337-43.
- [CrossRef] [PubMed] [Google Scholar]
- Fine-needle aspiration findings in nodular myositis: A case report. Diagn Cytopathol. 2000;23:343-7.
- [CrossRef] [Google Scholar]
- Role of FNAC in the diagnosis of skin and subcutaneous lesions. Int J Med Res Prof. 2019;5:272-9.
- [Google Scholar]
- The Cytology of Soft Tissue Tumours. In: Vol. Vol 16. Berlin, Germany; Karger; 2003.
- [CrossRef] [Google Scholar]
- Role of fine needle aspiration cytology in the initial diagnosis of superficial soft tissue lesions. Int J Contemp Med Res. 2018;5:C1-6.
- [Google Scholar]
- Diagnostic accuracy and limitations of fine-needle aspiration cytology of bone and soft tissue lesions: A review of 1114 cases with cytologicalhistological correlation. Cancer Cytopathol. 2010;118:24-32.
- [CrossRef] [PubMed] [Google Scholar]
- Scope of FNAC in the diagnosis of soft tissue tumors--a study from a tertiary cancer referral center in India. Cytojournal. 2007;4:20.
- [CrossRef] [PubMed] [Google Scholar]
- Role of fine needle aspiration cytology in the diagnosis of soft tissue tumours and tumour-like lesions. Cytopathology. 2004;15:32-7.
- [CrossRef] [PubMed] [Google Scholar]
- Enzinger and Weiss's Soft Tissue Tumors (6th ed). Philadelphia, PA: Saunders; 2014.
- [Google Scholar]
- Benign soft-tissue tumors in a large referral population: Distribution of specific diagnoses by age, sex, and location. AJR Am J Roentgenol. 1995;164:395-402.
- [CrossRef] [PubMed] [Google Scholar]
- MRI findings of subcutaneous epidermal cysts: Emphasis on the presence of rupture. AJR Am J Roentgenol. 2006;186:961-6.
- [CrossRef] [PubMed] [Google Scholar]
- Lever's Histopathology of the Skin (10th ed). Philadelphia, PA: Lippincott Williams and Wilkins; 2009.
- [Google Scholar]
- Infections of the musculoskeletal system by M tuberculosis In: Rom WN, Garay SM, eds. Tuberculosis (4th ed). New York: Little Brown and Company; 1996. p. :635-44.
- [Google Scholar]
- Fine-needle aspiration cytology in the diagnosis of tuberculous lesions. Lab Med. 2000;31:625-32.
- [CrossRef] [Google Scholar]
- Ganglion of the foot and ankle: Imaging and pathological findings, differential diagnosis, and operative management. J Orthop Res Physiother. 2015;1:1-4.
- [CrossRef] [Google Scholar]
- Hookworm-related cutaneous larva migrans acquired in the UK. BMJ Case Rep. 2015;2015:bcr2015210165.
- [CrossRef] [PubMed] [Google Scholar]
- Four years retrospective study of skin adnexal tumors: Histomorphology and special stain study. Int J Med Sci Public Health. 2020;9:273-9.
- [CrossRef] [Google Scholar]






