Translate this page into:
Survival rate of cervical cancer from a study conducted in India
*Corresponding author: Ganesh Balasubramaniam, Department of Medical Records Biostatistics and Epidemiology, Tata Memorial Centre, Centre for Cancer Epidemiology, Navi Mumbai, Maharashtra, India. drbganeshtmh@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Balasubramaniam G, Gaidhani RH, Khan A, Saoba S, Mahantshetty U, Maheshwari A. Survival rate of cervical cancer from a study conducted in India. Indian J Med Sci 2021;73(2):203-11.
Abstract
Objectives:
The incidence and mortality rates of cervical cancer in the world are 13.1/105 and 6.9/105, respectively. In India, it is estimated that there are 96,922 new cervical cancer cases and 60,078 deaths and ranked second among women cancers. The incidence and mortality rates are 14.7/105 and 9.2/105, respectively, although the incidence varies within Indian population. The majority of these cervical patients report at a late-stage of disease in health-care facility due to lack of awareness. A premier tertiary cancer hospital in Mumbai conducted the present survival-study. The aim of the study was to assess the various factors that determine the survival outcomes by age at diagnosis, role of comorbidities, stage of disease, and treatment.
Material and Methods:
The main source of information was patients’ medical records from which the data were abstracted, and active follow-up was undertaken. 1678 cases diagnosed between the years 2006 and 2008 at Tata Memorial Hospital are analyzed using actuarial method for obtaining survival rates and log-rank tests for comparison of survival groups.
Results:
The 5-year survival rate was 76.0% for those aged ≤50 years, 73.3% for non-residents, 74.4% for literates, and 81.8% for Christians and 72.5% for those with squamous cell carcinoma histology; those with no comorbidities had marginally better survival than their counter-parts. The survival rates were 83.5%, 80.6%, 66.0%, and 37.1% for Stage I, Stage II, Stage III, and Stage IV, respectively. The best survival outcomes were for those treated with only surgery (95.6%), or in combination with either radiotherapy (90.6%) or with radiotherapy and chemotherapy (85.5%). Involvement of either nodes/sites had poorer survival than those with no-involvement. In the multivariate analysis, only stage of the disease emerged as a significant prognosis factor.
Conclusion:
The study concluded that younger patients, early stage of disease, non-involvement of any sites/ nodes, and radiotherapy either alone or in combination with other treatment type provided better outcomes. Early detection and prevention strategies are keys to obtain better outcomes.
Keywords
Cervical cancer
Survival
India
Stage of disease
Treatment
INTRODUCTION
As per the GLOBOCAN 2018, there are an estimated 18,078,957 new cancers cases and 9,555,027 cancer deaths in the world with an incidence rate of 197.9 and mortality of 101.1 per 100,000.[1] Among women, cervical cancer was second most common and seventh among both the sexes, with an estimated 569,847 (3.2%) new cases in 2018 in the world; Asia alone accounted for 55.3% (n = 315,346 cases), and mortality accounted for 54.1% (168,411 deaths) of cervical cancer deaths. Cervical cancer is ranked 9th, in incidence as well as in mortality. The world age-standardized incidence and mortality rates for cervical cancer are 13.1/105 and 6.9/105, respectively.[1] In India, it is estimated that there are 96,922 new cervical cancer cases (9.2%) with an age-standardized incidence rate of 14.7/105 (higher than the rates observed in many other countries across the globe) and 60,078 cervical cancer deaths (8.4%) with a mortality rate of 9.2/105.[1] The incidence rates of cancer cervix within India showed variation. The incidence rates for cervical cancer in major Indian cancer registries are, 15.3 in Bengaluru (2012), 16.1 in Barshi (2012–2014), 15.9 in Chennai (2012–2013), and 19.0 in Mumbai (2012).[2]
Tata Memorial Hospital (TMH), Mumbai, India, a premier cancer institute is a tertiary cancer center in India. Annually, out of 45,000 new cancers seen, approximately 800–1000 new cervix cancers are diagnosed, of which about 75% undergo complete treatment at TMH. The voluminous data accrued offers opportunity to evaluate and assess outcomes in terms of survival rates out of clinical trials.
MATERIAL AND METHODS
The main source of information was patients’ medical records from which the data were abstracted. Classification[3] for site and morphology, and Federation of Gynecology and Obstetrics (FIGO) system[4] for staging of disease were used. The present study is a retrospective study comprising of histologically proven cervical cancer diagnosed between 2006 and 2008, in TMH. In all, 2428 cases were diagnosed and treated of which 1678 were treatment naive before registration at TMH and thus were considered as “Eligible” for this study. All patients underwent initial evaluation, staging work-up, histological confirmation, and joint clinic multi-disciplinary evaluation and treatment decision. The patient characteristics including demographic details, co-morbidities, tumor characteristics, treatment details including surgery, radiation, and chemotherapy were compiled. Active follow-up was undertaken periodically, if not then either telephonically or sending an email to those who did not attend for follow-up. House-visits were also undertaken for patients residing in Mumbai. Actuarial method[5] was applied to obtain survival rates and log-rank tests[6] to compare the survival rates between the groups. Statistical software IBM SPSS statistics for windows, version 21.0. Armonk, NY: IBM Corp. was used for statistical analysis.
Ethical approval
Ethical approval is obtained and the number is IRB CRA-320.
RESULTS
In all 1678 cases of cervical cancer were analyzed and distributions by general and clinical characteristics are shown in Table 1.
| General | Number of cases | Percent | Clinical | Number of cases | Percent |
|---|---|---|---|---|---|
| Total cases | 1679 | Stage | |||
| Age(in years) | I | 217 | 12.9 | ||
| <50 | 673 | 40.0 | II | 568 | 33.8 |
| >50 | 1006 | 60.0 | III | 768 | 45.7 |
| Residence | IV | 115 | 6.8 | ||
| Resident | 400 | 23.8 | Unk | 11 | 0.7 |
| Non-resident | 1278 | 76.2 | Complete treatment | 1562 | 93.1 |
| Marital status | Treatment type* | ||||
| Unmarried | 2 | 0.1 | S | 66 | 4.2 |
| Married | 1252 | 74.6 | R | 578 | 37 |
| Widow | 425 | 25.3 | C | 14 | 0.9 |
| Education | S+R | 42 | 2.7 | ||
| Illiterate | 856 | 51.0 | S+C | 19 | 1.2 |
| Literate | 823 | 49.0 | R+C | 792 | 50.7 |
| Religion | S+R+C | 51 | 3.3 | ||
| Hindu | 1444 | 86.1 | Type of surgery | ||
| Muslim | 133 | 7.9 | Total (extra-fascial) Abdominal hysterectomy | 20 | 1.2 |
| Christian | 27 | 1.6 | Modified radical Hysterectomy | 6 | 0.4 |
| Sikh | 61 | 3.6 | Radical abdominal hysterectomy | 179 | 10.7 |
| Jain | 14 | 0.8 | Extended radical Hysterectomy | 2 | 0.1 |
| Hypertension | Pelvic exenteration | 1 | 0.1 | ||
| No | 1396 | 83.2 | Others | 23 | 1.4 |
| Yes | 216 | 12.9 | Total | 231 | 13.8 |
| Diabetes | Radiation details | Tele-therapy | Brachytherapy | ||
| No | 1500 | 89.4 | Radiotherapy dosage (cGy) | No. of cases | No. of cases |
| Yes | 112 | 6.7 | 01–1000 | 72 | 42 |
| AID/HIV | 1001–2000 | 52 | 353 | ||
| No | 1555 | 92.7 | 2001–3000 | 78 | 551 |
| Yes | 55 | 3.3 | 3001–4000 | 181 | 99 |
| Histology | 4001–5000 | 982 | 8 | ||
| Squamous cell carcinoma | 1501 | 89.5 | 5001–6000 | 61 | 6 |
| Adenocarcinoma | 98 | 5.8 | 6001–7000 | 13 | 0 |
| Others | 80 | 4.8 | 7001–8000 | 2 | 0 |
| Subtotal | 1441 | 1059 |
The median age was 53 years. 76.2% were non-residents, outside of Mumbai, 74.6% married, 49% literate, and 86.1% Hindus, followed by 7.9% Muslims. With regard to the comorbidities, 12.9%, 6.7%, and 3.3% had history of hypertension, diabetes, and acquired immunodeficiency syndrome/human immunodeficiency virus (AIDS/HIV), respectively.
Tumor and treatment characteristics are shown in Table 1. The majority of the patients were squamous cell carcinoma (89.5%) and adenocarcinoma was 5.8%. The distribution by stage of disease was 12.9%, 33.8%, 45.7%, and 6.8% in Stage I, Stage II, Stage III, and Stage IV, respectively; Stage II and Stage III together accounted for almost 80%. About 93.1% of patients completed the full initial cancer-directed treatment and only 6.9% did not complete the treatment. Either radiotherapy alone or in combination with chemotherapy constituted a major proportion of treatment given to patients. It is seen from Table 1 that 37.0% were treated with radiotherapy alone and 50.7% were treated with radiotherapy in combination with chemotherapy. About 13.8% (231 cases) were treated surgically, of which 10.7% (179 cases) underwent “Radical abdominal hysterectomy.”
Of those treated with radiotherapy, 1441 cases were treated with “Tele-therapy” and 1051 with “Brachytherapy.”
Table 2 gives the survival rates (%), according to general characteristics. It is seen that those aged below 50 years had better survival, than those aged above 50 years. Patients who resided outside of Mumbai had marginally better survival than Mumbai. The differences in survival rates between illiterates and literates were less, and though there were differences in survival rates between religious groups, it was not statistically significant; marital status did show statistical difference, though there were only two cases who were unmarried.
| Characteristics | No. of cases | Survival rates (%) | |||||
|---|---|---|---|---|---|---|---|
| 1-year | 2-year | 3-year | 4-year | 5-year | P-value | ||
| All cases | 1679 | 92.5 | 83.8 | 78.0 | 74.7 | 72.6 | |
| ≤50 | 673 | 92.6 | 84.6 | 79.2 | 76.7 | 76.0 | 0.140 |
| >50 | 1005 | 92.4 | 83.2 | 77.1 | 73.2 | 70.1 | |
| Residence | |||||||
| Resident | 400 | 87.4 | 78.0 | 73.5 | 70.8 | 70.4 | 0.006 |
| Non-resident | 1278 | 94.2 | 85.7 | 79.4 | 76.0 | 73.3 | |
| Marital status | |||||||
| Unmarried | 2 | 50.0 | 50.0 | 50.0 | 50.0 | 50.0 | 0.003 |
| Married | 1252 | 93.8 | 86.0 | 79.7 | 76.7 | 74.6 | |
| Widow | 425 | 90.2 | 78.1 | 73.4 | 68.7 | 66.6 | |
| Education | |||||||
| Illiterate | 856 | 92.2 | 82.7 | 76.2 | 72.1 | 70.4 | 0.104 |
| Literate | 823 | 92.9 | 84.7 | 79.5 | 76.8 | 74.4 | |
| Religion | |||||||
| Hindu | 1444 | 92.5 | 83.9 | 77.9 | 75.0 | 73.0 | 0.470 |
| Muslim | 133 | 89.9 | 79.4 | 77.1 | 70.9 | 66.9 | |
| Christian | 27 | 96.1 | 91.8 | 87.2 | 87.2 | 81.8 | |
| Sikh | 61 | 96.2 | 84.6 | 74.2 | 68.5 | 68.5 | |
| Jain | 14 | 100.0 | 90.5 | 90.5 | 79.8 | 79.8 | |
| Hypertension | |||||||
| No | 1396 | 92.2 | 83.7 | 78.0 | 75.2 | 73.0 | 0.639 |
| Yes | 216 | 94.5 | 83.6 | 76.5 | 70.1 | 68.6 | |
| Diabetes | |||||||
| No | 1500 | 92.4 | 83.6 | 77.7 | 74.6 | 72.5 | 0.746 |
| Yes | 112 | 94.2 | 84.9 | 78.3 | 72.7 | 71.1 | |
| Acquired immunodeficiency syndrome/human immunodeficiency virus | |||||||
| No | 1555 | 92.4 | 83.9 | 77.8 | 74.5 | 72.4 | 0.947 |
| Yes | 55 | 96.0 | 79.8 | 77.3 | 74.8 | 70.8 | |
| Histology | |||||||
| Squamous cell carcinoma | 1501 | 92.8 | 84.3 | 78.3 | 74.8 | 72.5 | 0.40 |
| Adenocarcinoma | 98 | 89.0 | 75.9 | 72.9 | 71.3 | 71.3 | |
| Others | 80 | 91.8 | 83.3 | 79.1 | 79.1 | 76.4 | |
The presence/absence of comorbidities (hypertension, diabetes, and AIDS/HIV) did not show any differences in survival rate, and also the outcomes based on histological types did not show any statistically significant differences in survival rates.
Figure 1 shows survival rates of clinically important variables. Stage of the disease was important factor that showed differences in survival rates. Survival rates decreased with the increase in severity of the disease, Stage-IV showing the least rates [Figure 1]. The 5-year survival rates were 84.4%, 80.3%, 65.9%, and 37.1% for Stage I, Stage II, Stage III, and Stage IV, respectively, and differences in survival rates between stage of disease were statistically highly significant.
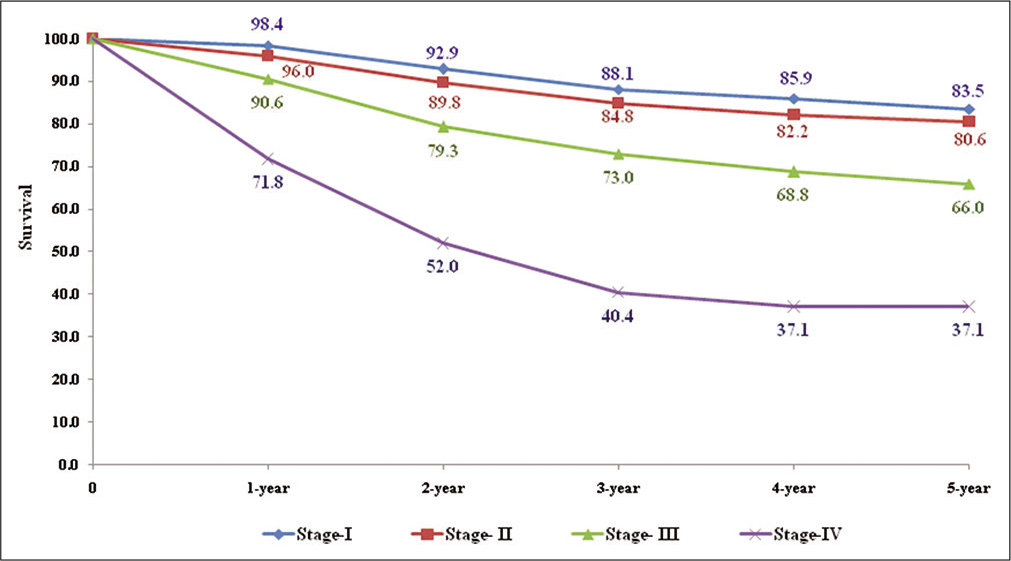
- Five-year observed survival rate (%) – by stage of disease.
Only cases, 1562 (93.1%), who completed treatment were considered for analysis. It may be seen from and Figure 2 that those treated with surgery alone (95.6%), or in combination with radiotherapy (90.6%) showed the best outcomes; those treated with surgery combined with chemotherapy (87.7%), or combined with both radiotherapy and chemotherapy (85.5%) showed similar outcomes but rates were lower when treated with radiotherapy combined with chemotherapy (76.6%); and the worst prognosis was for those treated with chemotherapy alone (15.9%). Radiotherapy was the most preferred treatment either alone or in combination with other treatment modalities. The differences in survival rates between treatments received were highly significant (P < 0.001).
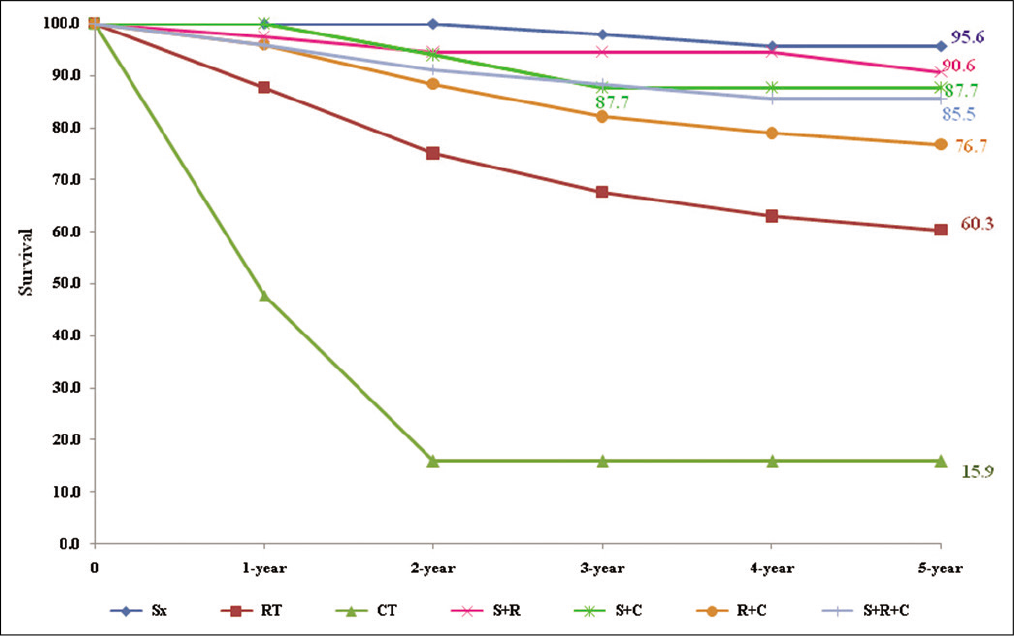
- Five-year observed survival rate (%) – by type of treatment.
Further it is shown that that the group “Any-surgery” (treated with surgery, either alone or in combination with other treatment modalities) showed 90.6%, 71.8% for “Any-radiotherapy” (treated with radiotherapy, either alone or in combination with other treatment modalities), and 76.8% for “Any-chemotherapy” (treated with chemotherapy, either alone or in combination with other treatment modalities) and the difference between these treatment groups were statistically significant.
Table 3 shows survival rates by Stage-treatment. In Stage I, rates for those treated with surgery alone (95.6%) were best, followed by those treated with surgery combined with radiotherapy (92.3%).
| Stage\treatment | No. of cases | Survival rates (%) | |||||
|---|---|---|---|---|---|---|---|
| 1-year | 2-year | 3-year | 4-year | 5-year | P-value* | ||
| Stage-I | |||||||
| S | 65 | 100.0 | 100.0 | 97.9 | 95.6 | 95.6 | <0.001 |
| R | 16 | 93.8 | 67.0 | 44.6 | 44.6 | 44.6 | |
| C | 0 | - | - | - | - | - | |
| S+R | 32 | 96.8 | 96.8 | 96.8 | 96.8 | 92.3 | |
| S+C | 4 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | |
| R+C | 19 | 100.0 | 94.6 | 77.9 | 72.3 | 66.3 | |
| S+R+C | 33 | 100.0 | 92.6 | 92.6 | 88.1 | 88.1 | |
| Stage-II | |||||||
| S | 0 | - | - | - | - | - | 0.002 |
| R | 143 | 93.2 | 82.8 | 74.9 | 70.6 | 67.1 | |
| C | 0 | - | - | - | - | - | |
| S+R | 10 | 100.0 | 83.3 | 83.3 | 83.3 | 83.3 | |
| S+C | 10 | 100.0 | 1000 | 88.9 | 88.9 | 88.9 | |
| R+C | 358 | 97.9 | 93.1 | 88.7 | 86.2 | 85.0 | |
| S+R+C | 15 | 86.7 | 86.7 | 78.4 | 78.4 | 78.4 | |
| Stage-III | |||||||
| S | 0 | - | - | - | - | - | <0.00 |
| R | 348 | 88.3 | 74.8 | 69.1 | 64.1 | 61.7 | |
| C | 5 | 50.0 | - | - | - | - | |
| S+R | 0 | - | - | - | - | - | |
| S + C | 3 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | |
| R+C | 377 | 94.6 | 85.1 | 78.4 | 74.3 | 71.1 | |
| S+R+C | 3 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | |
| Stage-IV | |||||||
| S | 0 | - | - | - | - | - | <0.001 |
| R | 68 | 64.0 | 51.2 | 36.6 | 18.3 | 18.3 | |
| C | 9 | 46.7 | 23.3 | 23.3 | 23.3 | 23.3 | |
| S+R | 0 | - | - | - | - | - | |
| S+C | 2 | 100.0 | 50.0 | 50.0 | 50.0 | 50.0 | |
| R+C | 33 | 86.4 | 64.8 | 49.3 | 49.3 | 49.3 | |
| S+R+C | 0 | - | - | - | - | - | |
Similarly in Stage II patients, it is seen that rates were highest for those treated with surgery combined with chemotherapy (88.9%), followed by and radiotherapy combined with chemotherapy (85%); the majority of cases in this stage were treated with radiation. In Stage III, the majority of patients treated with either only radiotherapy or combined with chemotherapy showed survival rates of 61.7% and 71.1%, respectively. In Stage IV, those treated with radiotherapy in combination with chemotherapy had the best survival rates (49.3%). There was statistical high significance in survival rates between treatments in each of the stages.
Involvement of some sites/nodes had an effect on the outcomes [Figure 3]. All surgically treated patients who had no involvement of uterus (92.5%), vagina (92.0%), parametrium (93%), ovaries (92%), regional nodes (94%), lymphovascular space (92%), and no-tumor-emboli (92%) showed better outcomes than those with respective involvements.
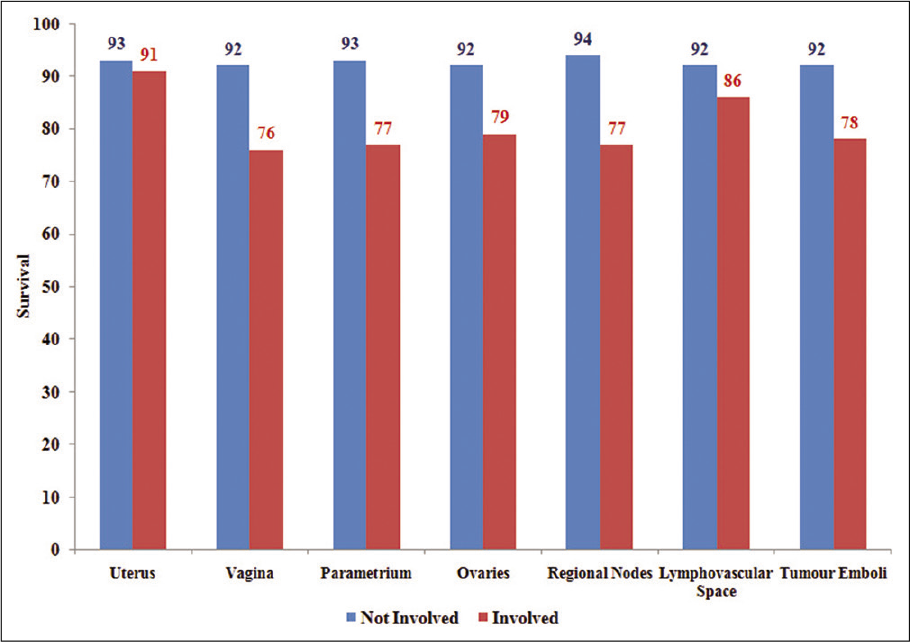
- Five-year observed survival rate (%) – by involvement of nodes/sites.
In a multivariate analysis, it is seen that none the factors, except stage of the disease, emerged as a significant prognostic factor [Table 4]; further, it is observed that risk, with Stage I as reference group, was 1.37, 2.27, and 3.07 for Stage II, Stage III, and Stage IV, respectively, and was statistically significant for Stage III and Stage IV; this implied that the risk increased with the severity, advancing of stage of disease.
| Characteristics | Risk | P-value | 95% CI (lower, upper) |
|---|---|---|---|
| Age (years) | |||
| ≤50 | Reference | ||
| >50 | 1.04 | 0.77 | (0.81, 1.3) |
| Place of residence | |||
| Mumbai | Reference | ||
| Others | 0.79 | 0.09 | (0.61, 1.04) |
| Marital status | |||
| Unmarried | Reference | ||
| Married | 2.30 | 0.59 | (0.11, 1.0) |
| Education | |||
| Literate | Reference | ||
| Illiterate | 0.80 | 0.17 | (0.7, 1.1) |
| Religion | |||
| Hindu | Reference | ||
| Muslim | 1.25 | 0.32 | (0.81, 1.91) |
| Others | .95 | 0.83 | (0.57, 1.57) |
| Hypertension | |||
| No | Reference | ||
| Yes | 1.29 | 0.16 | (0.9, 1.83) |
| Diabetes | |||
| No | Reference | ||
| Yes | 0.90 | 0.68 | (0.55, 1.48) |
| Acquired immunodeficiency syndrome/human immunodeficiency virus | |||
| No | Reference | ||
| Yes | 1.06 | 0.87 | (0.55, 2.02) |
| Histology | |||
| Squamous cell carcinoma | Reference | ||
| Adenocarcinoma | 1.28 | 0.34 | (0.77, 2.13) |
| Others | 0.80 | 0.49 | (0.43, 1.49) |
| Stage of disease | |||
| I | Reference | ||
| II | 1.37 | 0.20 | (0.85, 2.2) |
| III | 2.27 | 0.00 | (1.44, 3.58) |
| IV | 3.07 | 0.00 | (1.70, 5.53) |
| Unknown | 0.89 | 0.92 | (0.10, 7.6) |
DISCUSSION
Cervical cancer is one of the leading cancer among women in India, accounting for 96,922 new cases and 60,078 deaths annually and contributes to one-fifth of women cancers in India.[1] The incidence in Asian population was higher than that seen in other regions of the world.[7] This disease is more prevalent in developing and under-developed countries, not so in the west.[8] In India, in early part of this century, though breast cancer was leading site of cancer in many of the registries, there were exceptions like Chennai among the urban and Barshi among the rural registries where cervix cancer was the leading site of cancer.[9]
The health infrastructure for early detection and treatment facilities in different populations has direct impact on the global variations of cervical cancer. About 30 population-based and six hospital-based cancer registries are setup by National Cancer Registry Programme (NCRP), a research unit under Indian Council of Medical Research (ICMR) and data from these registries provide statistics on cancer in India. The NCRP initiated and started a National project for common cancers in India as head and neck, breast, and cervix cancer, in which a core-pro forma was developed and tested for data collection, and it included details as, demography, comorbidities, clinical stage, pathological stage, treatment details, and its complications supplemented with periodical follow-up. This data are used for evaluating the patterns of care and survival outcomes. The data used for this study are from patients who attended the hospital for diagnosis and treatment. There are already published studies reported on cervical cancer survival outcomes from India. Much of the focus has been to improve the survival duration and quality of life with less toxicity and side effects of treatment through conduct of clinical trials. There have been remarkable improvements in cervical cancer management.
One of the earliest studies on cervical cancer survival reported in 1998 from Mumbai outlined on observed and relative survival rates by stage of disease.[10] In this study, the 5-year overall observed and relative survival rate was 51.0% and 54.8%, respectively. In the present study, the 5-year observed that survival rates by stage of disease were 84.4%, 80.3%, 65.9%, and 37.1% for Stage I, Stage II, Stage III, and Stage IV, respectively, and were comparable to outcomes as poorer survival for advanced stage diseased patients.[10] These results of Mumbai study were comparable to some of the earlier studies published from India.[11-15]
Subsequent to this, a study conducted in Mumbai assessed the survival outcomes of cervical cancer by age, religion, education, and extent of disease.[16] The 5-year relative survival rate for younger patients (<35 years) was 47.4% but decreased with increasing age, unmarried had a better survival, and Muslims had a better survival than Hindus, though education did not have any impact on prognosis. Survival decreased with advancing clinical extent of disease, 5-year rates of 69.3% for localized cancer, 41.6% for regional spread, and less than 5% for distant metastasis. As envisaged, age at diagnosis and clinical extent of disease emerged as significant factors in a multivariate analysis.
In comparison to the above study, in the present study, Christians showed better survival rates than other religious groups and literates had better survival outcomes than illiterates, and both were not statistically significant. Those younger than 50 years had better survival outcomes than above the age of 50 years. The differences in the survival outcomes may be explained by the fact the present study is hospital-based and has patients from various parts of India, while the Mumbai study is a population-based study. None of the comorbidities as hypertension, diabetes, and AIDS/HIV history showed any statistical difference in survival rates in the present study.
An international publication based on survival data from various countries, SURVCAN, especially Asian countries was published by international agency for research on cancer in the year 2010.[17] In this report, the 5-year relative survival rates reported by Indian population registries were 46.4%, 59.6%, 34.5%, 35.7%, and 57.8% in Mumbai, Chennai, Bhopal, Barshi, and Karunagapally, respectively, though it was 46%, overall, in India. The rates in India, among the Asian countries, were lower than that reported in other Asian countries as China (67%), Singapore (66%), Turkey (63%), South Korea (79%), and Thailand (61%). This clearly indicates that there are disparities in survival rates within the country (like India), and also between countries (Asian).[17]
According to hospital based cancer registry of TMH, in 2006– 2008 cervical cancer was the second leading site of cancer among females.[18] The majority of the patients completed the initial planned treatment, but 75% had a follow-up, either as a demographic follow-up or clinical follow-up. It is recognized that with the advancement of stage of disease, the follow-up rate decreased, probably as there were many patients residing outside of the city and were from rural parts of State of Maharashtra.
Earlier, in a large retrospective study (1979 – 1994) cervical cancer cases treated with radical radiation, comprising of 1891 of Stage IIb and 3321 of Stage IIIb patients showed an 8-year disease-free survival of 56% and 40% for Stage IIb and Stage IIIb, respectively.[19]
The improvement in outcomes over time may be due to down-staging and improved compliance for completion of treatment and follow-up. Furthermore, approximately 30–35% of patients participated prospective clinical trials where outcome tends to be better as compared to routine care.
Difference in outcomes by histology was reported in a study conducted in the rural part of India.[20] In this study, it was shown that the survival outcomes were better for patients with squamous cell carcinoma than adenocarcinoma, which was also observed in the present study. The 5-year survival rate 95.1% and 5.3% for Stage IA and stage IV, respectively, compared to 83.5% and 37.1% for Stage-I and Stage-IV, respectively, in the present study. In the present study, it was observed that when radiotherapy was added to surgery as a treatment combination, the prognosis improved in all the stages and a similar outcome was reported when intracavitary radiotherapy was given as a combination treatment in another study on a screening population in rural India.[20]
To assess factors that affect prognosis, cervical cancer diagnosed in a cohort of a screening-trial study was reported.[21] The 5-year overall observed survival rate was 32.5%, 78% in Stage I and 9% in Stage IV patients, and a 70% higher risk of death for those with poor socio-economic status; however, it reported that higher household income was significantly associated with poorer survival. However, in the present study, information on income is not reliable since the hospital where the study is conducted treats 60% patients free of cost.
A review of studies on gynecological cancer covered a wide plethora of issues as demographics, pathology, clinical findings, presence of HPV affecting the outcomes, and screening trial conducted in India, and a host of studies reported.[22] One of the issues that was mentioned was the difference in staging of cancers in the SEER and FIGO as used by the clinicians and might have an impact on reporting of survival outcomes.
A retrospective study on locally advance cervical cancer conducted in a premier tertiary cancer hospital, recently, reported that in patients who were treated with complete definitive or adjuvant radiotherapy, survival rate was 62% for Stage II and 45% for Stage III patients, and use of concurrent radiotherapy as 32% and 57% for CTRT, and the resultant outcomes were comparable to those reported in other parts of the world.[23]
The American society published 5-year relative survival rates of cervical cancer patients seen between 2009 and 2015;[24] the rates were 66% for all SEER Stages combined, 92% for localized, 56% for regional, and 17% for distant-metastasis patients.
To compare survival outcomes in different countries, CONCORD study based on 279 population-based cancer registries in 67 countries reported cancer survival of cases registered between 1995 and 2009.[25] The global 5-year survival was less than 40% and more than 70% indicating a wide range. There was a decline in survival over the period in France which they attributed to intensive screening for pre-invasive lesions, resulting in removal of less aggressive tumors;[26,27] however, in Nordic countries, the survival was stable same or increased.[28] In the CONCORD study, cervical cancer survival rates were greater than 50% in many countries, except in Benghazi, Libya (39%), and Karunagappally, India (46%). In countries with lower/ middle-income, the survival rates were low since invasive cervical cancer is potentially curable with early detection by screening and appropriate surgery.[29]
A similar study like the present study was conducted in Khon-Kaen, Thailand and reported by Sriamporn et al. (2004).[30] In this study, those who were aged <40 years had best survival and those above 60 years had the poorest survival, proving an inverse relationship of age and survival, similar to the present study outcomes, that is, those less than 50 years to have better prognosis than those above the age of 50 years. The 5-year survival rate was 74.6% and 38.2% for Stage I and Stage (III + IV), respectively, and is comparable to present study rates (Stage I: 83.5%, Stage III: 66%, and Stage IV: 37.1%). Survival rates by involvement in the present study showed only parametrium involvement and regional nodes involvement as significant factor in univariate analysis, which was similar to a study reported elsewhere.[31] Nonetheless, the survival rates have been better for those with is no involvement of sites/nodes.
Outcomes based on tumor size was published in a recent study in (2017) from Martinique registry (French-West Indies) known for high cervical cancer incidence;[32] it was reported that those <60 years (65%), Stage (I + II) patients (71%), and tumor size less than 4 cm (85%) had better survival outcomes.
In cervix cancer, it is known that radiotherapy is one of the major treatment modality and the outcomes have been very good in terms of survival. In an earlier study, it was reported that the cumulative 5-year survival rates for Stage Ib, Stage II, and Stage III/IVa were 93.5%, 77.0%, and 60.3%, respectively, which are very good especially in view of Stage IV patients treated with radiotherapy;[33] the outcomes are similar in present study as well.
To summarize the present retrospective study, marital status, stage of the disease, treatment, outcomes of stage by treatment, involvement of regional nodes, and laterality of positive nodes/parametrium were of significance for the outcomes in terms of survival. These variables are of prime importance and are taken into account for treatment management. However, it is to be mentioned that the hospital where the study has been conducted, has shown a stage-shift in cervical, meaning that a decade back, major proportion of cervical cancer patients used to report in either Stage III or Stage IV disease; this phenomenon has changed, and the hospital sees more cases in Stage II and Stage III. Keeping in view of this, and also the technology advancement for diagnosis and targeted therapy, the outcomes have improved over a period of time. There are some strengths and limitations of the present study. The strengths are the cohort of patients who completed treatment capturing the treatment modalities and analyzing outcome, being an hospital-based study; the limitations being that it is a retrospective study and thus outcomes may be assessed and not the treatment effect on survival like in a clinical trial.
CONCLUSION
In the past two decades, with the availability of newer facilities there has been a significant refinement in the treatment protocols and outcome. This was possible due to multidisciplinary approach in the management of cervical cancer. For patients with early or advanced disease, treatment with surgery/radiotherapy/chemotherapy or a combination of these treatment modalities will be based on clinical, radiological imaging, and pathology findings. Early detection improves the survival rate of cervical cancer. The present study clearly shows that radiotherapy administered either alone or in combination with surgery or chemotherapy has better prognosis. The findings from various studies re-iterates the need for formulating strategies to increase awareness of cancer so that patients arrive at health facilities at an early stage of disease and provides opportunity for clinicians to provide best treatment. The key to improve survival outcomes is awareness, screening programs, and providing the optimal treatment. Due to the lack of cancer awareness, limited health-care facilities available in developing countries, such as India, most women report at advanced stage of diseases, which adversely affects the prognosis and thus focus to improve these will be the key for better outcomes.
ACKNOWLEDGMENTS
The authors would like to be thank to Dr. R A Badwe, Director for providing his support and encouragement, (NCRP now National centre for disease informatics and research), ICMR for collaboration. The authors would like to thank and acknowledge Mrs. Shubha Kothare and (late) Mrs. Vidya Lanke for their contribution in data abstraction. Special thanks to Mrs. Mitali Sapkal and Mrs. Sharwari Joshi for the data entry and patient’s follow-up activities. Also, the authors would like to thank and acknowledge all the staff of all the departments of the hospital.
Declaration of patient consent
Institutional Review Board permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- International Agency for Research on Cancer. 2018. GLOBOCAN 2018 Database. Lyon, France: International Agency for Research on Cancer; Available from: http://www.gco.iarc.fr [Last accessed on 2020 Jul 02]
- [Google Scholar]
- Three-Year Report of Population Based Cancer Registries 2012-2014. Incidence, Distribution, Trends in Incidence Rates and Projections of Burden of Cancer (Report of 27 PBCRs in India) 2016. National Centre for Disease Informatics and Research, National Cancer Registry Programme, Indian Council of Medical Research. Available from: http://www.ncdirindia.org/NCRP/all_ncrp_reports/pbcr_report_2012_2014/all_content/pdf_printed_version/preliminary_pages_printed.pdf [Last accessed on 2020 Jul 02]
- [Google Scholar]
- International Classification of Diseases for Oncology (3rd ed). Geneva: World Health Organization; 2013.
- [Google Scholar]
- Revised 2018 International Federation of Gynecology and Obstetrics (FIGO) cervical cancer staging: A review of gaps and questions that remain. Int J Gynecol Cancer. 2020;30:873-8.
- [CrossRef] [PubMed] [Google Scholar]
- Calculation of survival rates for cancer. Proc Staff Meet Mayo Clin. 1950;25:270-86.
- [Google Scholar]
- Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep. 1966;50:163-70.
- [Google Scholar]
- Cancer Incidence in Five Continents Vol 11. Lyon: International Agency for Research on Cancer; 2017.
- [Google Scholar]
- GLOBOCAN 2008 Cancer Incidence and Mortality Worldwide: IARC Cancer Base No. 10 Lyon, France: International Agency for Research on Cancer; 2010.
- [Google Scholar]
- Two-Year Report of the Population Based Cancer Registries 2006-2008 Bangalore, India: National Cancer Registry Programme, Indian Council of Medical Research; 2009.
- [Google Scholar]
- Long-term survival from uterine cervical cancer in Mumbai (Bombay), India. Int J Cancer. 1998;78:394-5.
- [CrossRef] [Google Scholar]
- Cervical cancer in Kerala: A hospital registry-based study on survival and prognostic factors. Br J Cancer. 1995;72:1039-42.
- [CrossRef] [PubMed] [Google Scholar]
- Global variations in cancer survival, Study group on cancer survival in developing countries. Cancer. 1996;78:2461-4.
- [CrossRef] [Google Scholar]
- Incidence, mortality and survival in cancer of the cervix in Bangalore, India. Br J Cancer. 1995;71:1348-52.
- [CrossRef] [PubMed] [Google Scholar]
- Cancer survival in Khon Kaen Province, Thailand. Int J Cancer. 1995;61:296-300.
- [CrossRef] [PubMed] [Google Scholar]
- Improved survival in cervical cancer cases in a rural Indian population. Br J Cancer. 1996;74:285-7.
- [CrossRef] [PubMed] [Google Scholar]
- Population-based survival from cancers of breast, cervix and ovary in women in Mumbai, India. Asian Pac J Cancer Prev. 2004;5:308-15.
- [Google Scholar]
- Cancer Survival in Africa, Asia, the Caribbean and Central America (Survcan) Lyon: IARC Scientific Publication, International Agency for Research on Cancer; 2010.
- [Google Scholar]
- Hospital Cancer Registry In: Annual Report-2006-2008. Mumbai: Tata Memorial Hospital; 2015.
- [Google Scholar]
- Treatment and outcome in cancer cervix patients treated between 1979 and 1994: A single institutional experience. J Cancer Res Ther. 2013;9:672-9.
- [CrossRef] [PubMed] [Google Scholar]
- Improved survival of cervical cancer patients in a screened population in rural India. Asian Pac J Cancer Prev. 2016;17:4837-44.
- [Google Scholar]
- Survival of patients with cervical cancer in rural India. J Clin Gynecol Obstetr. 2015;4:290-6.
- [CrossRef] [Google Scholar]
- Gynecological cancers: A summary of published Indian data. South Asian J Cancer. 2016;5:112-20.
- [CrossRef] [PubMed] [Google Scholar]
- Locally advanced cervical cancer: A study of 5-year outcomes. Indian J Cancer. 2018;55:45-9.
- [CrossRef] [PubMed] [Google Scholar]
- American Cancer Society. Last Medical Review. Available from: https://www.cancer.org/cancer/cervical-cancer/detection-diagnosis-staging/survival.html [Last accessed on 2020 Jan 03]
- [Google Scholar]
- Global surveillance of cancer survival 1995-2009: Analysis of individual data for 25,676,887 patients from 279 population-based registries in 67 countries (CONCORD-2) Lancet. 2015;385:977-1010.
- [CrossRef] [Google Scholar]
- Survival of cancer patients in France: A population-based study from the association of the French Cancer Registries (FRANCIM) Eur J Cancer. 2007;43:149-60.
- [CrossRef] [PubMed] [Google Scholar]
- Variations in survival for invasive cervical cancer among European women, 1978-89. EUROCARE working group, Cancer Causes Control. 1999;10:575-81.
- [CrossRef] [PubMed] [Google Scholar]
- Trends in the survival of patients diagnosed with cancer in female genital organs in the Nordic countries 1964-2003 followed up to the end of 2006. Acta Oncol. 2010;49:632-43.
- [CrossRef] [PubMed] [Google Scholar]
- Expansion of cancer care and control in countries of low and middle income: A call to action. Lancet. 2010;376:1186-93.
- [CrossRef] [Google Scholar]
- Loss-adjusted survival of cervix cancer in Khon Kaen, Northeast Thailand. Br J Cancer. 2004;91:106-10.
- [CrossRef] [PubMed] [Google Scholar]
- Lymph nodes metastasis in cervical cancer: Incidences, risk factors, consequences and imaging evaluations. Asia Pac J Clin Oncol. 2018;14:e380-5.
- [CrossRef] [PubMed] [Google Scholar]
- Epidemiology and survival of cervical cancer in the French West-Indies: Data from the Martinique cancer registry (2002-2011) Glob Health Action. 2017;10:1337341.
- [CrossRef] [PubMed] [Google Scholar]
- Treatment of squamous cell carcinoma of the uterine cervix with radiation therapy alone: Long-term survival, late complications, and incidence of second cancers. Br J Cancer. 2007;97:1058-62.
- [CrossRef] [PubMed] [Google Scholar]






