Translate this page into:
COVID-19 in people living with HIV – A case series
*Corresponding author: Bhavya Chadalavada, Department of Internal Medicine, Gandhi Medical College, Secunderabad, Telangana, India. bhavi1794@hotmail.com
-
Received: ,
Accepted: ,
How to cite this article: Chadalavada B, Rao MR, Pandharpurkar D. COVID-19 in people living with HIV – A case series. Indian J Med Sci 2022;74:99-102.
Abstract
People with chronic diseases, old age, obesity, and immunosuppression are more susceptible to infections, including COVID-19. Human immunodeficiency virus (HIV) is widely prevalent in India, yet, the number of COVID cases in people living with HIV is low. We report six cases of COVID-19 in patients with HIV who face various challenges in the intensive care unit. The severity of their COVID infection varies from patient to patient depending on their immune status and treatment with antiretroviral therapy.
Keywords
HIV
COVID-19
Tuberculosis
Critical care
Highly active antiretroviral therapy
INTRODUCTION
HIV infection is widely prevalent in India, yet, the number of COVID cases in people living with HIV (PLHIV) is low, and not many cases series/reports have been published. PLHIV who are not on treatment or have treatment failure are more at risk of contracting the coronavirus.
CASE SERIES
Case 1
A 30-year-old male from Telangana reported shortness of breath, low-grade fever, myalgias, dry cough, and increasing chest discomfort. On a positive COVID reverse transcription-polymerase chain reaction (RT-PCR), he was transferred to Gandhi Hospital. The patient is a known case of HIV, on Antiretroviral therapy-Tenofovir/Lamivudine/Efavirenz regimen since 2016 but incompliant, current CD4 count 139 and viral load not detected. At the time of admission, the patient was dyspneic at rest with a respiratory rate of 33 breaths/min, blood pressure of 120/80 mm Hg, pulse rate of 103 beats/min, and saturation of 86% at room air, and body temperature of 100°F. On day 1, a chest X-ray (CXR) revealed right upper lobe consolidation with bilateral basal infiltrates. High-resolution computed tomography (HRCT) CHEST showed bilateral diffuse ground-glass opacification predominantly in the peripheries and consolidation in the right upper lobe. On day 2 of admission, antiretroviral therapy (ART) was changed to T. Tenofovir/Lamivudine 300/300 mg combination once daily and T. Lopinavir/Ritonavir 200/50 mg twice daily following reports that protease inhibitors had a better outcome in COVID patients.[1] Sputum for cartridge-based nucleic acid amplification test (CB-NAAT) tested positive on D10. He was started on anti-tubercular therapy with rifabutin in place of rifampicin and monitored. Sodium level persistently remained below 130, urinary sodium was 50, and urine osmolality was 250. After ruling out, all other causes of hyponatremia Syndrome of Inappropriate Antidiuretic Hormone secretion was suspected. Sodium levels improved after fluid restriction. On day 15, fever spikes reduced and the patient appeared comfortable with room air. He was slowly weaned off oxygen support. The patient was discharged on D20 and asked for a follow-up in the outpatient department.
Case 2
A 47-year-old male, plumber by occupation, person living with HIV for 14 years on Zidovudine/Lamivudine/ Nevirapine regimen, complained of high-grade fever, dry cough, and shortness of breath on exertion for 4 days. He was referred to Gandhi hospital due to suspicion of COVID. On examination, pulse rate (PR) was 100 beats/min, respiration rate (RR) 31 breaths/min, oxygen saturation 64% at room air, and blood pressure (BP) 130/80 mmHg. CXR showed B/L infiltrates. Nasopharyngeal swab for RT-PCR was positive. CD4 count was 109 and viral load 90,000. On day 3, the patient’s saturation dropped and he was shifted to noninvasive ventilation. Ventilator settings were kept at Fi02 80% and PEEP 15 cm H20 on continuous positive airway pressure (CPAP) mode. On day 4, the patient’s ART regimen was changed to Tenofovir, lamivudine, and dolutegravir (TLD) in view of the treatment failure. After 8 days of intensive care unit (ICU) stay, we were able to wean him off CPAP and onto an oxygen mask. On day 17, he was comfortable at room air, 6-min walk test did not show any drop in saturation. He was shifted to the ward from the ICU.
Case 3
A 55-year-old male, unemployed, from Hyderabad, presented to our hospital with progressive shortness of breath and high-grade fever for 4 days. Patient was COVID RT-PCR positive. On further probing into his history, he hinted at a chronic disease for which he never took treatment. On examination, the patient was conscious, coherent, poorly built, and emaciated, BP 110/80 mg, PR 94 beats/min, RR 38 breaths/ min, Temp 99.4, and SpO2 86% at room air. He was started on oxygen inhalation, IV antibiotic, IV steroid, and Inj Remdesvir. Routine laboratories were done were normal. HIV serology was positive and CD4 count was 89. Arterial blood gas (ABG) showed PO2 86 mmHg on 8 L of oxygen, PCO2 30.1, and pH 7.44. PaO2/FiO2 was 191. The patient’s respiratory distress worsened over the next 3 days with a fall in pO2 to 40 mmHg on 100% FiO2 on CPAP mode. He was intubated but went into cardiac arrest during the procedure and could not be revived.
Case 4
A 58-year-old male with a known history of diabetes for 5 years and hypertension for 3 years presented with low-grade fever, productive cough, and progressive shortness of breath for 10 days. On examination, the patient was drowsy but arousable, tachypneic, BP 90/60 mmHg, PR 113 beats/min, RR 32 breaths/min, Temp 101.4, and SpO2 68% at room air. He was immediately started on O2 inhalation and IV antibiotic, IV fluid, and IV paracetamol. SARS-Cov-2 RT-PCR was positive. CXR showed bilateral diffuse infiltrates with lower lobe predominance. HRCT chest showed multiple ground-glass opacifications with a CT severity score of 25/25. Incidentally, HIV was positive. From day 2, ABG showed a worsening of PaO2/FiO2 ratio despite oxygen support. We started him on IV steroid and IV Remdesvir. On day 3, the patient was put on CPAP ventilation at Positive End Expiratory Pressure 10 cm H20 and 60% FiO2. He became stuporous on the same day. Blood sugars, serum electrolytes, and CT brain were normal. During intubation, the patient went into cardiac arrest and could not be revived.
Case 5
A 36-year-old male, farmer by occupation with no known comorbidities, presented to the hospital with complaints of low-grade fever for 5 days, sudden onset shortness of breath, sudden onset pain in the epigastric and right hypochondrium, right-sided chest pain, and aggravated on inspiration. On examination, the patient was conscious, coherent, BP 110/80 mmHg, PR 100 beats/min, RR 22 breaths/min, Temp 99.4, and SpO2 93% at room air. Auscultation revealed reduced breath sounds in the right mammary, infra axillary, and infrascapular areas. CXR showed an air-fluid level in the right upper zone suggestive of hydropneumothorax, a thin-walled cavity in the apex of the right lung. CT chest done confirmed hydropneumothorax with multiple loculations. SARS-Cov-2 RT-PCR was positive. HIV Elisa was positive and CD4 count was 360. We did a tube thoracostomy and 750 ml of pus was collected immediately. This aspirate showed lymphocytic predominance. The patient’s respiratory effort reduced once the drain was inserted and he was continued on oxygen support. Collection from the drain was 200 ml on day 2. From days 3–5, 20–30 ml pus was collected and collection stopped thereafter. The patient received IV antibiotics, analgesics, and antipyretics. On day 2, the patient recorded low blood pressure and was started on inotrope, which gradually tapered as his blood pressure improved by day 3. On day 4, sputum for CB-NAAT returned positive. The patient was started on anti-tuberculosis treatment (ATT). Repeat CXR after 1 week showed no collection in the right lung, chest drain was removed. After 20 days of hospital stay, the patient tested COVID negative and was planned for discharge. He was advised to follow up after 4 weeks to start highly active antiretroviral therapy (HAART).
Case 6
A 34-year-old male, driver by occupation, was admitted to the hospital with a history of headache, multiple episodes of vomiting, high-grade fever, and involuntary micturition for 5 days. Following admission, the patient had one episode of right-sided focal seizures with impaired awareness history of significant weight loss of 20 kg in 6 months. On examination, patient was conscious, drowsy, BP 110/90 mmHg, RR 18/ min, and SpO2 96% at room air. On doing a neurological examination, patient was irritable and not oriented to time and place. Cranial nerve examination was normal. Motor examination revealed right hemiparesis, increased tone, and exaggerated reflexes on the right. Neck rigidity was present. On routine examination, incidentally, human immunodeficiency virus enzyme-linked immunoassay HIV ELISA was positive, COVID RT-PCR was positive. CD4 count was 122. CXR showed the right upper zone fibrosis and perihilar lymphadenopathy. Magnetic resonance imaging brain showed the left frontoparietal space-occupying lesion with perilesional edema and midline shift as shown in Figures 1 and 2. Contrast Enhanced Computed Tomography brain showed multiple peripherally enhancing conglomerate isodense lesions in the left frontal lobe with mass effect suggestive of brain abscess. A lumbar puncture was done and cerebrospinal fluid (CSF) was sent for analysis – 13 cells, all lymphocytes, proteins 70 mg/ dl, glucose 26 mg/dl, gram stain, and culture not significant, CSF for CBNAAT was positive. On day 4 of admission, the patient was started on ATT and IV steroids for tubercular meninogoencephalitis with the left frontal tubercular abscess. Fever spikes and headache subsided, sensorium improved on day 15. We continued monitoring him in the ward for the next week. On day 22, the patient was discharged and asked to review after 2 weeks to start HAART.

- MRI brain showing left frontal lobe heterogenous cystic lesion iso-intense on T2 FLAIR with extensive oedema and mass effect.

- MRI brain showing left frontal lobe heterogenous cystic lesion hypointense on T1 with extensive oedema and mass effect.
RESULTS
Here, we have reported six different cases of PLHIV with COVID-19 infection and their progression which have been summarized in table 1 shown above and Figure 3 below. Case 1 was a diagnosed PLHIV non-compliant with medication; Case 2 was a diagnosed PLHIV on the same regimen for 14 years, rest of the four patients were de novo diagnosed with HIV in our hospital. The severity of their infection and outcome also greatly varied depending on their immune status and treatment with ART. Patients 1 and 2 who were on ART were treated and discharged albeit some complications during their hospital stay. Patient 1 on the TLE regimen was shifted to protease inhibitor-containing regimen TLATv/R. Patient 2 was in treatment failure and the regimen changed to TLD. Patients 3 and 4 who were de novo diagnosed with HIV developed severe respiratory failure and did not survive. Patients 5 and 6 had associated tuberculosis and were started on ATT before ART. Among the patients with HIV-TB coinfection, patient 1 had pulmonary TB, patient 5 had pulmonary TB with hydropneumothorax, and patient 6 had TB meningitis with tubercular abscess.
| Case 1 | Case 2 | Case 3 | Case 4 | Case 5 | Case 6 | |
|---|---|---|---|---|---|---|
| Sex | Male | Male | Male | Male | Male | Male |
| Age | 30 | 47 | 55 | 58 | 36 | 34 |
| HIV Status | Known PLHIV | Known PLHIV | De novo HIV | De novo HIV | De novo HIV | De novo HIV |
| Opportunistic Infections | Pulmonary Tuberculosis | Pulmonary Tuberculosis with hydropneumothorax | TB meningitis with Tubercular abscess | |||
| Comorbidities | DM, HTN | |||||
| CD4 Count | 139 | 109 | 89 | 360 | 122 | |
| Outcome | Discharged | Discharged | Death | Death | Discharged | Discharged |
PLHIV: People living with HIV, DM: Diabetes mellitus, HTN: Hypertension
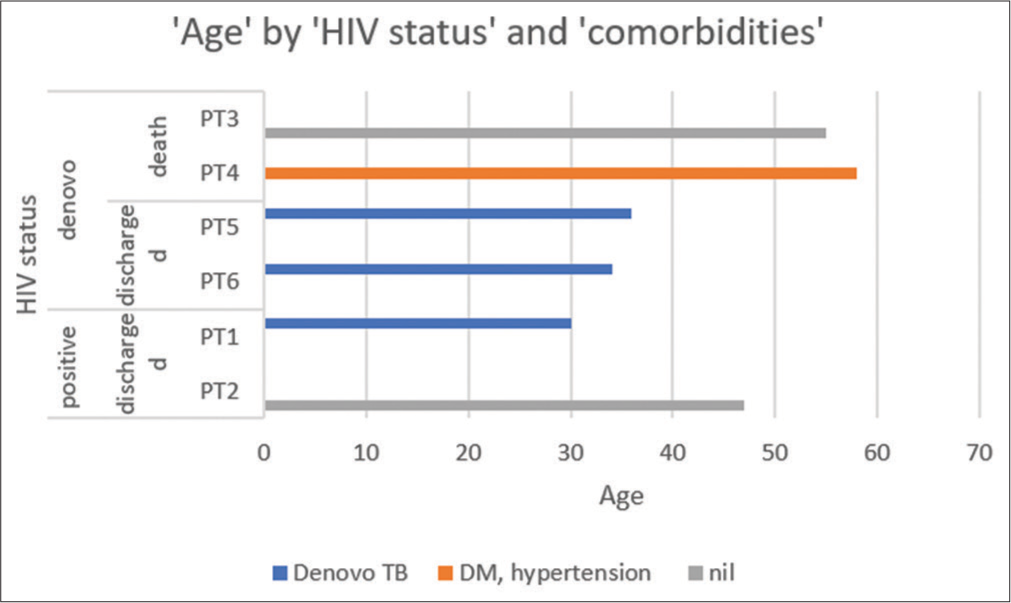
- Distribution of patients by age, HIV status, comorbidities and hospital outcomes.
DISCUSSION
Suwanwongse and Shabarek observed the clinical features and outcomes of nine HIV/SARS-CoV-2 coinfected patients and found that low CD4+ T cell counts in the HIV-positive patients may adversely affect the COVID-19 outcomes.[2] Another study by Maggiolo et al. compared 55 cases of SARSCoV-2 infection with 69 asymptomatic PLHIV negative for SARS-CoV-2 RT-PCR and suggested that HIV-positive individuals are not protected from SARS-CoV-2 infection or have a lower risk of severe disease and those with low CD4+ T cell counts might have worse outcomes.[3]
Several clinical features, old age, elevated inflammatory markers, presence of comorbidities, and CXR/HRCT abnormalities are predictive factors for severe COVID-19 and we found a similar pattern in HIV/COVID-19 coinfected patients. It is clear from our case series that HIV-related immunosuppression does not have any protection from COVID-19 infection. PLHIV not on HAART and with low CD4 count is more likely to manifest a severe respiratory disease[2] if infected by COVID. On the other hand for PLHIV, CD4 count of more than 200 (source: BHIVA recommendation), undetectable viral load,[4] and compliance with HAART, they might be at no greater risk than the general population.[5]
It is recommended that to be safe during this pandemic, all PLHIV with a suppressed viral load change their current HIV regimen. They should be categorized as an extremely vulnerable group and follow strict social distancing protocol, stay at home preferably, and avoid socializing. They should also get priority access to COVID-19 vaccination. All HIV/SARS-COV-2 coinfected people should receive prophylaxis against opportunistic infections. Screening for TB in PLHIV and vice versa is also necessary.[6] More research is required to learn the management of COVID infection in an immunosuppressed individual.
CONCLUSION
Initiation, ensuring compliance with HAART and identifying treatment failure is essential to prevent severe COVID related illness in PLHIV during this pandemic.
Declaration of patient consent
Patient consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Repurposing antiviral protease inhibitors using extracellular vesicles for potential therapy of COVID-19. Viruses. 2020;12:486.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical features and outcome of HIV/SARS-CoV-2 coinfected patients in The Bronx, New York city. J Med Virol. 2020;92:2387-9.
- [CrossRef] [PubMed] [Google Scholar]
- SARS-CoV-2 infection in persons living with HIV: A single center prospective cohort. J Med Virol. 2021;93:1145-9.
- [CrossRef] [PubMed] [Google Scholar]
- COVID-19 infection in a HIV positive health care worker: First case report from a tertiary care hospital of North India. Virusdisease. 2021;32:320-4.
- [CrossRef] [PubMed] [Google Scholar]
- Impact of COVID-19 on people with HIV: A review. J Virus Eradic. 2020;6:100019.
- [CrossRef] [PubMed] [Google Scholar]
- HIV testing among patients with presumptive tuberculosis: How do we implement in a routine programmatic setting? Results of a large operational research from India. PLoS One. 2016;11:e0156487.
- [Google Scholar]






